Aniline CAS Number: 62-53-3
Basic Information
Aminobenzene,
aniline
, anilin
, anilin oil
, aminobenzene,
anilin
aniline 038-01[6]
Aminobenzene, anilin,
aniline, β-eucaine
aniline
, 99+% aniline, ACS, 99+%
AKOS BBS-00003680
AMINOBENZENE
ANILINE
ANILINE OIL
BENZENAMINE
BENZENEAMINE
PHENYLAMINE
ai3-03053
amino-benzen
Aminophen
Anilin
anilin(czech)
Anilina
anilina(italian,polish)
Aniline reagent
aniline(andhomologs)
Aniline(benzenamine)
aniline15
anilineandhomologues
Anyvim
Pesticide intermediates: Fungicide intermediates: Other types of fungicides
Physicochemical properties
Security Data
very toxic hazardous materials
Acute Tox. 3 Inhalation
Acute Tox. 3 Oral
Aquatic Acute 1
Aquatic Chronic 1
Carc. 2
Eye Dam. 1
Muta. 2
Skin Sens. 1
STOT RE 1
Application areas
It is one of the most important intermediates in the dye industry, and also a major raw material for pharmaceuticals, rubber accelerators, and antioxidants. It can also be used to make fragrances, varnishes, and explosives.
It is used as an analytical reagent, and also in the synthesis of dyes, resins, lacquer, and fragrances.
Aniline is an important raw material for the production of pesticides. Aniline can be used to derive N-alkylaniline, alkylaniline, o-nitroaniline, o-phenylenediamine, phenylhydrazine, cyclohexylamine, etc. It can be used as an intermediate for fungicides such as sodium chlorothalonil, seed dressing agent, methyl thiophanate, thiophanate-methyl, carbendazim, pyrimethanil, and benomyl; insecticides such as triazophos, pyridazin, and quinalphos; and herbicides such as metolachlor, acetochlor, butachlor, cycloazinone, and imidazoline.
Aniline is an important intermediate. There are over 300 major products derived from aniline. There are approximately 80 aniline producers worldwide, with a total annual production capacity exceeding 2.7 million tons per year and an output of about 2.3 million tons. The main consumption sector is MDI, which accounted for approximately 84% of total aniline consumption in 2000. In my country, aniline is mainly consumed in MDI, dyes, rubber additives, pharmaceuticals, pesticides, and organic intermediates. In 2000, aniline consumption was 185,000 tons, insufficient to meet demand, necessitating imports. Aniline intermediates and dyes include: 2,6-diethylaniline, N-acetaniline, p-butylaniline, o-phenylenediamine, diphenylamine, diazoaminobenzene, 4,4′-diaminotriphenylmethane, 4,4′-diaminodiphenylcyclohexylmethane, N,N-dimethylaniline, N-ethylaniline, N,N-diethylaniline, N,N-dipropylaniline, p-acetamidophenol, p-aminoacetophenone, 4,4′-diethylaminobenzophenone, 4-(p-aminophenyl)butyric acid, and p-nitrobenzene. Amines, N-nitrosodiphenylamine, β-acetanilide, 1,4-diphenylaminourea, 2-phenylindole, p-phenylaminoaniline, N-formylaniline, N-benzoylaniline, N-acetaniline, 2,4,6-trichloroaniline, p-iodoaniline, 1-aniline-3-methyl-5-pyrazolone, hydroquinone, dicyclohexylamine, 2-(N-methylanilino)propionitrile, 3-(N-ethylanilino)propionitrile, 2-(N-ethylanilino)ethanol, p-aminoazobenzene, phenylhydrazine, monophenyl Urea, diphenylurea, p-thiocyanoaniline, 4,4′-diphenylmethane diisocyanate, polyphenyl polymethyl polyisocyanate, 4-aminoacetaniline, N-methyl-N-(β-hydroxyethyl)aniline, N-methyl-N-(β-chloroethyl)aniline, N,N-dimethyl-p-phenylenediamine, N,N,N’,N’-tetramethyl-p-phenylenediamine, N,N-diethyl-p-phenylenediamine, 4,4′-methylenebis(N,N-diethylaniline), phenylthiourea, diphenylthiourea, p-aminobenzenesulfonic acid, 4,4′-Diaminodiphenylmethanebenzoquinone, N,N-diethanolaniline, acetoacetaniline, p-aminophenol, N-ethylbenzylaniline, N-methylformylaniline, N-methylacetoacetaniline, p-bromoacetoacetaniline, bis(p-aminocyclohexyl)methane, phenylhydrazone diphenylcarbazone, acetophenone phenylhydrazone, aniline-2,4-disulfonic acid, p-aminoazobenzene-4′sulfonic acid, phenylhydrazine-4-sulfonic acid, thioacetoacetaniline, 2-methylindole, 2,3-dimethylindole, N-methyl-2-phenylindole
It is one of the most important intermediates in the dye industry. It is also a major raw material for pharmaceuticals, rubber accelerators, and antioxidants; it can be used to make fragrances, varnishes, and explosives, and is also used as a solvent. It is used in ICP-AES, AAS, AFS, ICP-MS, ion chromatography, etc. It is a standard solution for titration analysis. It is used for calibrating instruments and apparatus; evaluation methods; working standards; quality assurance/quality control; and other applications.
Preparation method
Aniline is currently produced by catalytic hydrogenation of nitrobenzene. The commonly used catalyst is Cu-SiO₂ , which exhibits good selectivity, readily reducing nitrobenzene to aniline while minimizing the risk of hydrogenation on the benzene nucleus. The reaction takes place in a fluidized bed reactor. Purified hydrogen is heated to 350–400°C and then enters the evaporator. Simultaneously, nitrobenzene enters the evaporator from a high-level tank, vaporizes upon contact with the hot hydrogen, and is superheated to 180–223°C. The mixed gas enters from the bottom of the fluidized bed and reacts with a copper catalyst supported on silica gel within the fluidized bed. The resulting crude aniline and water vapor are discharged from the top of the bed. The crude aniline is cooled and separated in a condenser, and then distilled to obtain the final product, aniline. Aniline production is now continuous, with the reduction reaction conducted under atmospheric pressure boiling reflux conditions, enabling large-scale production with small equipment.
The main industrial production methods for aniline include the nitrobenzene iron powder reduction method, the chlorobenzene amination method, the nitrobenzene catalytic hydrogenation reduction method, and the phenol ammonolysis method. Among these, the chlorobenzene amination method has been completely discontinued abroad due to high costs and equipment corrosion. The nitrobenzene hydrogenation reduction method is currently the main production method used both domestically and internationally. A few foreign companies, such as Mitsui Petrochemicals (30,000 t/a unit) in Japan and Steel Chemicals (90,000 t/a unit) in the United States, still use the phenol amination process. Others use the traditional iron powder reduction method, such as Bayer in Germany and Mobel in the United States, which utilizes byproduct iron oxide to co-produce iron-based raw materials, thus making the overall economic benefits superior to the hydrogenation method. 1. Iron Powder Reduction Method: Nitrobenzene is reduced with iron powder. The reaction solution is neutralized with lime, washed, and then distilled to obtain the finished product. 2. Hydrogenation Reduction Method: Nitrobenzene undergoes gas-phase hydrogenation reduction in a fluidized bed reactor in the presence of a copper catalyst to obtain crude aniline. The reaction solution is condensed and separated into layers, then distilled under reduced pressure to obtain the finished product.
Upstream and downstream product information
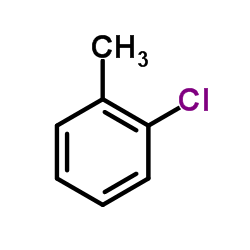
8-Aniline-1-naphthalenesulfonic acid Direct Orange S Pigment Red 208 2,2,4-Trimethyl-1,2-dihydroquinoline Polymer Pigment Red 175 3-Bromo-2-pyridinecarboxylic acid Fluorescent whitening agent N,N-dicyanoethylaniline 4-decylaniline 4-Bromo-2-trifluoromethylquinoline Golden Red Aniline Hydrochloride Reactive Blue 222 Reactive Violet 5 Dissolving Salt SV Dicyclohexylamine 2-chloromalealdehyde N-phenylpyridine-4-amide Acid Yellow 79 Acid Black 26 Lidamidine Basic Orange 1-Naphthylaminobenzene 4-hydroxy-2-trifluoromethylquinoline Aniline Black Dispersible Red S-3GFL 1,3-Diphenylurea N-phenyl-2- naphthylamine Acid Black 234 Wool Reactive Blue PW-3R Direct Dark Brown NM Direct Maroon NGB Direct Dark Green 2G m-Hydroxydiphenylamine Phenyhydrazine Sulfate 2,4,6-Trichloroaniline N-Phenylethanol Liquefied MDI Sudan-1 N,N’-Diphenyl-1,4-phenylenediamine
Material Safety Data Sheet (MSDS)
62-53-3 (Safety characteristics, toxicity, storage and transportation)
It can explode when mixed with air; it reacts violently with oxidizers.
The warehouse should be well-ventilated, kept at a low temperature and dry; it should be stored separately from oxidants and food additives.
Skin – Rabbit 20 mg/24 hours: Moderate; Eye – Rabbit 20 mg/24 hours: Moderate
Highly toxic
Oral LD50 in rats: 250 mg/kg; Oral LD50 in mice: 464 mg/kg
Open flame, high temperature, strong oxidizers are flammable; high heat decomposition produces toxic nitrogen oxide gases.
Toxic substances
Foam, carbon dioxide, dry powder
TLV-TWA 2 PPM; TWA 5 PPM (19 mg/m³); STEL 20 mg/m³
Frequently Asked Questions List
Aniline is the simplest primary aromatic amine, a compound formed by replacing one hydrogen atom in a benzene molecule with an amino group. It is a colorless, oily, flammable liquid with a strong odor. Its melting point is -6.3℃, boiling point is 184℃, relative density is 1.0217 (20/4℃), refractive index is 1.5863, flash point (open cup) is 70℃, autoignition point is 770℃, and it decomposes at 370℃. It is slightly soluble in water and readily soluble in organic solvents such as ethanol, ether, and chloroform. It turns brown upon exposure to air or sunlight. It can be distilled using steam, with a small amount of zinc powder added during distillation to prevent oxidation. Purified aniline can be treated with 10–15 ppm of NaBH4 to prevent oxidation and deterioration.
Aniline solutions are alkaline and readily form salts with acids. The hydrogen atom on its amino group can be replaced by a hydrocarbon or acyl group to form secondary or tertiary anilines and acylanilines. Substitution reactions primarily produce ortho- and para-substituted products. Aniline reacts with nitrous acid to form diazonium salts, from which a series of benzene derivatives and azo compounds can be produced.

Aniline is an important chemical raw material, with over 300 major products manufactured. It is mainly used in MDI, the dye industry, pharmaceuticals, and as a rubber vulcanization accelerator, such as p-aminobenzenesulfonic acid in the dye industry and N-acetanilide in the pharmaceutical industry. It is also a raw material for manufacturing resins and coatings. In 2008, aniline consumption was approximately 360,000 tons, and the estimated demand in 2012 was approximately 870,000 tons, while the commercial aniline production capacity reached 1.37 million tons, resulting in an overcapacity of nearly 500,000 tons. Aniline is highly toxic to blood and nerves, and can cause poisoning through skin absorption or inhalation.
Industrially, aniline is mainly produced using two methods:
1. Hydrogenation of nitrobenzene via active copper catalysis; this method allows for continuous production and is pollution-free.
2. Reaction of chlorobenzene and ammonia at high temperature in the presence of a copper oxide catalyst.
This information was edited and compiled by Xiaonan of ChemicalBook (2020-02-06).
In recent years, there has been considerable research in my country on the synthesis of aniline from the reaction of benzene with H₂O₂. In 2003, Xiamen Advanced Technology Co., Ltd. developed a process for the direct synthesis of aniline from benzene, ammonia, and oxygen. This process features high benzene conversion and good selectivity. Aniline is synthesized from benzene with 25% ammonia and oxygen at 1.5 MPa and 140–160 °C, with a total selectivity of 94% (plus approximately 5% phenol and 1% benzene). If aniline is synthesized from benzene, liquid ammonia, and oxygen at 2.0–8.1 MPa and 160–200 °C, the total selectivity is 95%. The catalysts used in this process include metal oxides, metal chlorides, metal fluorides, metal iodides, metal sulfates, metal phosphates, metal heteropolyacids, BF3, metals other than iron powder, and mixtures of the above compounds in any proportion. The support is at least one metal or non-metal compound selected from SiO2, B2O3, Al2O3, GeO2, TiO2, ZrO2, Nb2O5, Ta2O5, activated carbon, etc.
In 2004, the College of Chemistry of Sichuan University and the Key Laboratory of Green Chemistry and Technology of Sichuan Province used H2O2 as an oxidant to achieve the direct oxidative amination of benzene to synthesize aniline, and disclosed a patent for the preparation method. Researchers have developed a Ni-Zr-Ce/Al2O3 catalyst. Under mild conditions of atmospheric pressure and 50℃, this catalyst has good activity for the direct oxidative amination of benzene, ammonia, and H2O2 to produce aniline. The selectivity for aniline production is much greater than that for phenol. Increasing the proportion of ammonia to benzene in the reaction feedstock can increase the yield of aniline without increasing the amount of phenol produced. This method boasts low energy consumption and high atom utilization, providing a new route for the green synthesis of aniline. The catalyst for the one-step synthesis of aniline is obtained through impregnation and calcination, using γ-Al₂O₃ as a support and two or three of nickel, molybdenum, vanadium, manganese, zirconium, and cerium as the active catalyst components. In the one-step synthesis of aniline from the direct oxidative amination of benzene, this catalyst exhibits high activity and up to 100% selectivity under relatively mild reaction conditions.
1. Aniline reacts with acetic acid under sulfuric acid catalysis (Experimental method: In a 50ml round-bottom flask, place 10ml of freshly distilled aniline, 15ml of glacial acetic acid, and a small amount of zinc powder. Attach a fractionating column, insert a thermometer, and reflux for about 1 hour. After the reaction is complete, stir and pour the hot reactants into a beaker containing 250ml of cold water. Cool and filter, wash the crude product, recrystallize the crude product, and filter again.) Generally, a fractionating column is used to remove water to shift the equilibrium and increase the yield: CH3COOH + C6H5NH2 → CH3CONHC6H5 + H2O.
2. Acetyl chloride reacts with aniline via ammonolysis (Experimental method: Add 5 drops of aniline to a dry test tube, then slowly add 8 drops of acetyl chloride. After the reaction is complete, add 5ml of water and stir with a glass rod) (Usually, a tertiary amine is added to neutralize the acid produced): CH3COCl + C6H5NH2 → CH3CONHC6H5 + HCl.
3. Ammonolysis of acetic anhydride in aniline (usually by adding a tertiary amine to neutralize the resulting acid): (CH3CO)O + C6H5NH2 → CH3CONHC6H5 + CH3COOH
Toxicity Protection
Extremely toxic. Inhalation of its vapors or absorption through the skin can cause poisoning, resulting in headache, dizziness, loss of appetite, increased pulse, vomiting, and hematuria; in severe cases, it can lead to death. Workplaces must be well-ventilated, and equipment must be sealed to prevent leaks, spills, and drips. Alcohol consumption can exacerbate poisoning. The oral LD50 for dogs is 300 mg/kg. The maximum permissible concentration in workplace air is 5 mg/m³. Operators must wear protective equipment, and are strictly prohibited from drinking alcohol before and after work, and must not eat or drink at the workplace.
Packaging and Storage
Packaged in airtight iron drums, each drum weighing 200 kg net. Store in a cool, well-ventilated place. Protect from moisture and sunlight during storage and transportation. Store and transport according to regulations for toxic and hazardous materials.
FAQ
Q1: About the after-sale service of products
A: After purchasing the products from our factory, we have A professional technical team and after-sales team to serve you and solve all your problems in the future.
Q2: Can I get some samples?
A: Yes, we can provide samples, but the customer will pay the freight.
Q3: How do I start paying?
Payment can be made by wire transfer or T/T, apple_pay, google_pay, gc_real_time_bank_transfer , etc.
Q4: How to confirm product quality before placing an order?
A: You can get free samples of some products. You just have to pay the shipping fee or arrange for the sample to be sent to us by express.
You can send us your product specifications and requirements and we will produce products according to your requirements.
Q5: What is your MOQ?
A: The minimum quantity we can order is 1kg.
But usually we can accept a smaller quantity, say 100g, at the cost of 100% sample charge.
Q6: Shipping Time?
A: We ship the parcel out in 1-2 days and offer tracking No.. Shipping time is different to different country. Please consult
Details:
| Item | Result |
| Appearance | liquid |
| Molecular formula | C4H11N |
| Boiling point | 36-38 °C |
| Molecular weight | 73.14 |
Usage:
Packaging and Shipping:
200L/drum
Storage:
Storage in ventilated low temperature dry warehouse; Keep container closed and separately from open flame and heat source

Methyl carbamate Basic introduction
1. Chemical Identity
- Common Name: Methyl carbamate
- IUPAC Name: Methyl carbamate
- CAS Number: 598-55-0
- It is the methyl ester of carbamic acid. It is structurally analogous to ethyl carbamate (urethane), but with a methyl group instead of an ethyl group.
2. Basic Properties
- Appearance: White crystalline solid at room temperature.
- Solubility: Soluble in water and common polar organic solvents like alcohols.
- Key Characteristic: Serves as the simplest member of the alkyl carbamate ester family, containing both amide and ester functional groups.
2. Methyl carbamate Primary Application Fields
Methyl carbamate is primarily an industrial chemical intermediate used in the synthesis of various high-value products. Unlike its more well-known analogue ethyl carbamate (urethane), it is not a significant natural contaminant and its uses are focused on active synthesis in several key industries.
1. Polymer and Chemical Synthesis
- Polymer Industry: Can be used as a monomer or chain modifier in the production of certain specialty polyurethanes and other resins. It helps impart specific properties like hardness or chemical resistance.
- Organic Synthesis: Serves as a protecting group for amines in multi-step synthetic routes. The carbamate group can be introduced to protect an amine from reacting under specific conditions and later removed (e.g., with acid hydrolysis).
2. Research and Development (R&D)
- Laboratory Chemical: Used in academic and industrial R&D as a standard reagent for synthesizing carbamate derivatives, studying reaction mechanisms, or developing new synthetic methodologies.


Methyl carbamate Basic introduction
1. Chemical Identity
- Common Name: Methyl carbamate
- IUPAC Name: Methyl carbamate
- CAS Number: 598-55-0
- It is the methyl ester of carbamic acid. It is structurally analogous to ethyl carbamate (urethane), but with a methyl group instead of an ethyl group.
2. Basic Properties
- Appearance: White crystalline solid at room temperature.
- Solubility: Soluble in water and common polar organic solvents like alcohols.
- Key Characteristic: Serves as the simplest member of the alkyl carbamate ester family, containing both amide and ester functional groups.
2. Methyl carbamate Primary Application Fields
Methyl carbamate is primarily an industrial chemical intermediate used in the synthesis of various high-value products. Unlike its more well-known analogue ethyl carbamate (urethane), it is not a significant natural contaminant and its uses are focused on active synthesis in several key industries.
1. Polymer and Chemical Synthesis
- Polymer Industry: Can be used as a monomer or chain modifier in the production of certain specialty polyurethanes and other resins. It helps impart specific properties like hardness or chemical resistance.
- Organic Synthesis: Serves as a protecting group for amines in multi-step synthetic routes. The carbamate group can be introduced to protect an amine from reacting under specific conditions and later removed (e.g., with acid hydrolysis).
2. Research and Development (R&D)
- Laboratory Chemical: Used in academic and industrial R&D as a standard reagent for synthesizing carbamate derivatives, studying reaction mechanisms, or developing new synthetic methodologies.























![CAS No.15875-13-5 Supply high quality 1,3,5-Tris[3-(dimethylamino)propyl]hexahydro-1,3,5-triazine /Best price/IN STOCK](https://cdn.globalso.com/mit-ivy/微信截图_20230425104915.png)